
The alpha-bromo compound series – Determining acidic or alkaline qualities
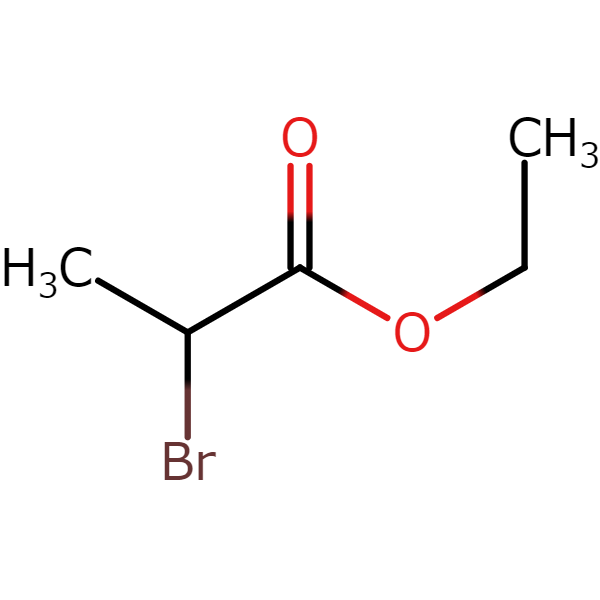
【CAS No.】535-11-5
【Chemical Name】Ethyl 2-bromopropionate
【Chemical formula】C5H9BrO2
Manac produces and markets compounds that contain halogen elements (halogen compounds), such as bromine and iodine, and are used as chemical intermediates in products such as medication, electronics, and semiconductor material. Examples of Manac’s product series include alkyl halides, alpha-bromo compounds, and heterohalogen compounds.
This article reviews the characteristics and applications of the alpha-bromo compound series.
■ What you can learn from this article ✔ Manac manufactures alpha-bromo compounds, which are mainly used as intermediates in pharmaceuticals and semiconductor materials. ✔ Alpha-bromo compounds are highly reactive and bond easily with other compounds using little energy. ✔ Manac excels in the production of brominated compounds and coupling reactions. ■ Recommended Articles ・ Making complex compounds a breeze! The heterohalogen series ・ Extensive use in electronic material intermediates and beyond! The alkyl halide series ・ MANAC’s three strengths in manufacturing active pharmaceutical ingredients and intermediates
contents
What are alpha-bromo compounds?
Alpha-bromo compounds are compounds formed by joining bromine (Br) with the carbon atom adjacent to the functional group (alpha carbon). Functional groups are structures that determine specific characteristics of compounds, such as acidic (propensity to release hydrogen ions) or alkaline (propensity to accept hydrogen ions) properties.
For example, compounds that contain a carboxylic acid group (-COOH) are acidic, while compounds with an amino group (-NH2) are alkaline. Compounds that contain an aldehyde group (-CHO) easily oxidate and become carboxylic acids.
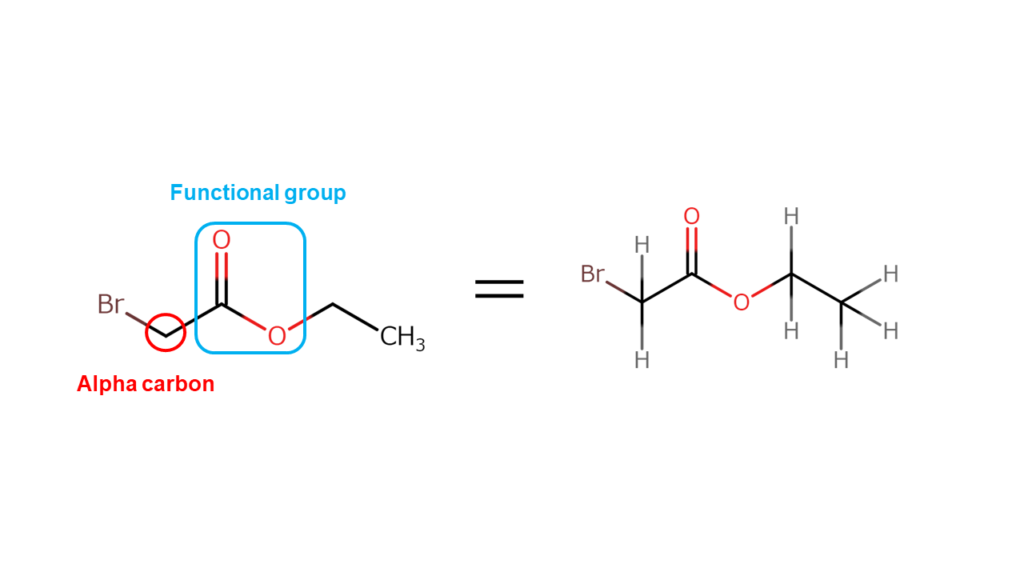
Compounds such as alpha-bromo compounds with a halogen bonded at the alpha position are generally highly reactive and may cause rashes or itchiness if it comes into contact with skin. Being able to handle such a dangerous substance on an industrial scale is a strength unique to Manac, with its extensive experience in handling bromine.
“Gluing” compounds together using halogen sites
Joining two types of compounds is a two-step process:
- Break the original bonds of the two compounds
- Create new bonds between the two compounds
Usually, a large amount of energy is required to break the original bonds of a compound, but because bonds between carbon and bromine are relatively weak, a well-placed application of a small amount of energy can break their bonds. This causes the bromine sites in the alpha-bromo compound to bond with other compounds (coupling reaction).
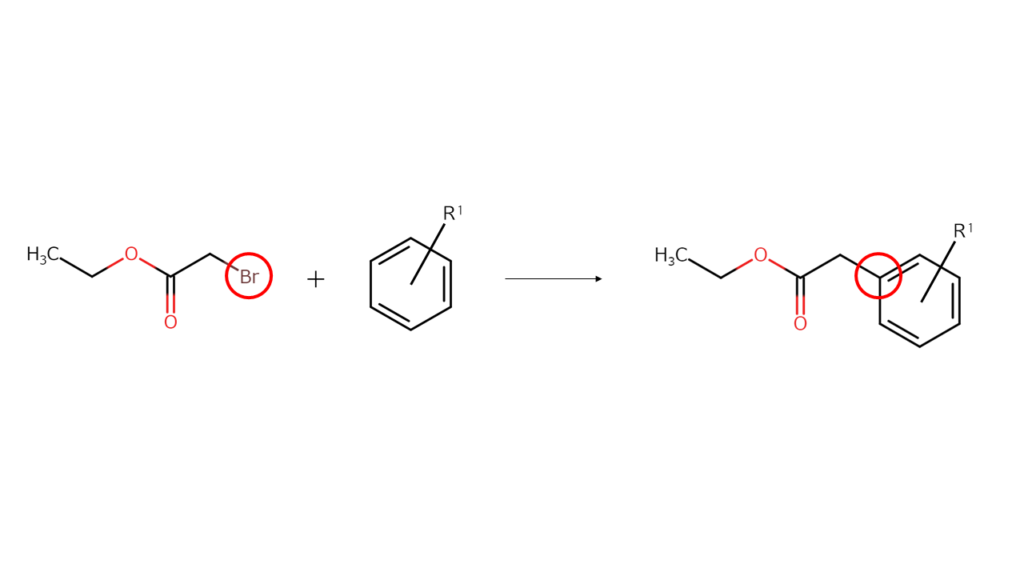
This means that the bromine present in alpha-bromo compounds acts as a “glue” to stick other compounds to its site.

Usage as “parts” for final compounds
The compounds (final compounds) used in electronic materials and medicines are generally of complex structures. In order to make final compounds, compound structures must be divided into several parts before joining the appropriate parts together. Alpha-bromo compounds are ideal for use as one of these parts with their halogen sites acting as the glue.
For example, let’s say we want to use compound A to make compound B, as shown below.
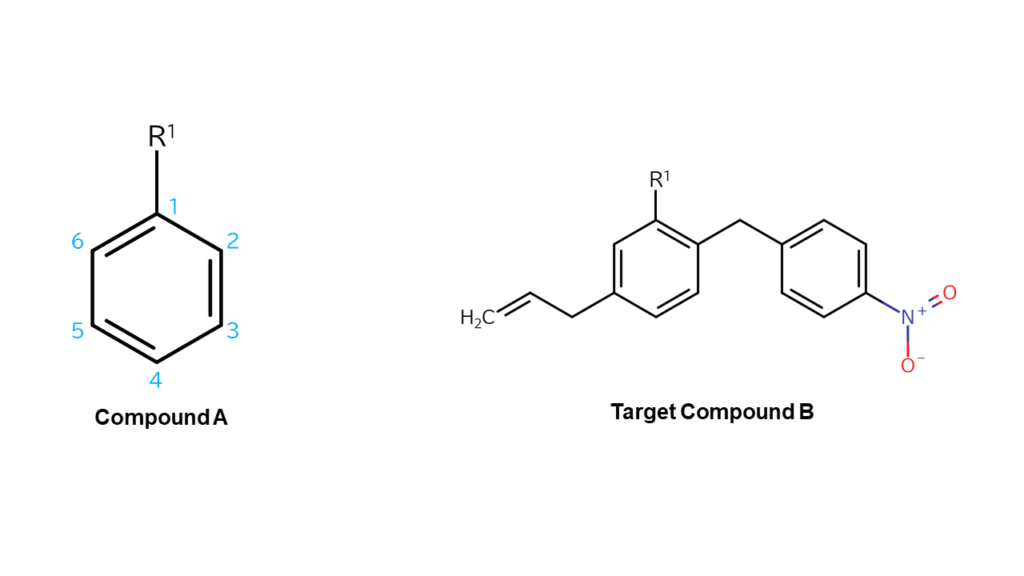
In order to make final compound B, we would need to bond the following structure to the second carbon of compound A:
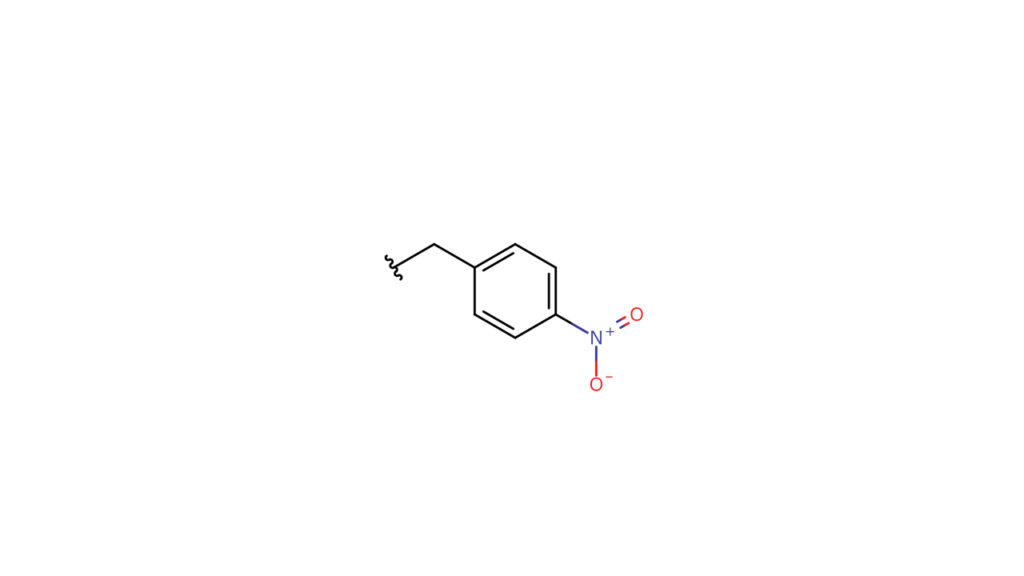
We would also need to bond the following structure to the fifth carbon of compound A:
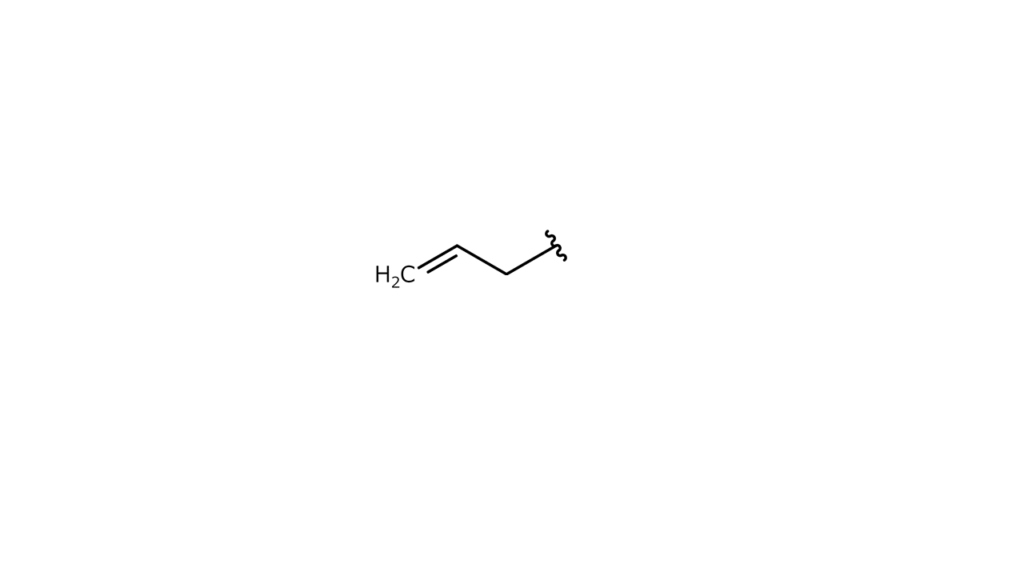
In this case, we would use compounds that are formed by bonding these structures with bromine (p-nitrobenzyl bromide and allyl bromide) as “parts.”
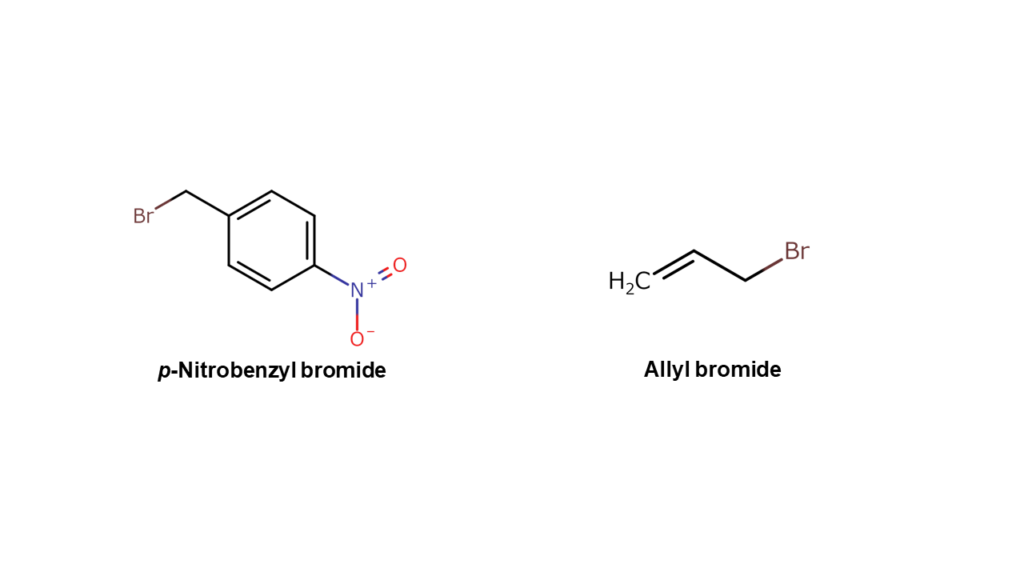
Causing a coupling reaction between these alpha-bromo compounds with compound A allows us to make final compound B.
Series list and applications of each alpha-bromo compound
There are many different compounds in Manac’s alpha-bromo compound series, which are mainly used as intermediates (parts) in products such as medicines and light-sensitive materials.
The following table lists the alpha-bromo compounds marketed by Manac, along with their applications and structural formulas. Be sure to check whether there are any “parts” in which you, the reader, may be interested.
Manac specializes not only in bromination and iodination to make alpha-bromo compounds, but also in coupling reactions of various types using alpha-bromo compounds. This enables Manac to handle complete orders, from manufacturing intermediates to producing higher-order compounds.
If you are interested in any of the parts in the above catalog, feel free to contact Manac for more details regarding higher-order compounds that are synthesized using those parts.














