Patent: Selectively manufacture needed compounds!
The “certain chemical reaction” that makes purification of iodoaniline mixtures possible
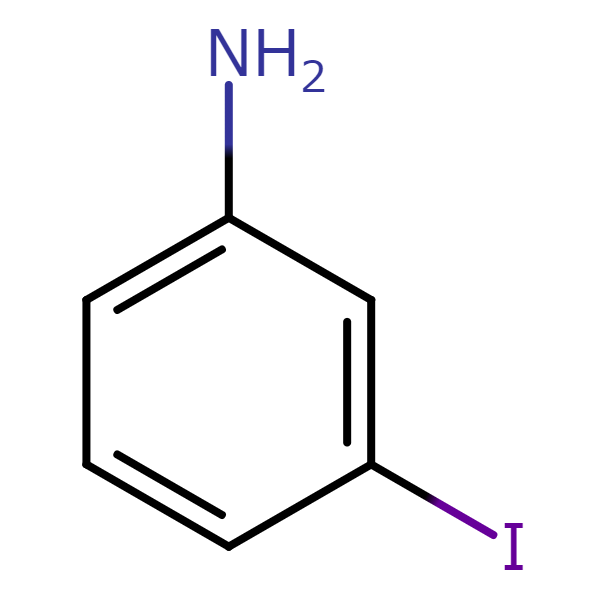
【CAS No.】626-01-7
【Chemical name】3-Iodoaniline
【Chemical formula】C6H6IN
Many chemical compounds (intermediates) are used in things we use every day, such as in electrical appliances and pharmaceuticals. These intermediates are essential for making electronics fire-resistant, including televisions, and making pharmaceuticals effective.
The functionality required of electronics and pharmaceuticals differs, so the structure of the intermediates used in them naturally differs. For this reason, it is crucial for manufacturers of chemical intermediates to accurately produce intermediates with the specified structure.
However, in some cases, compounds with similar structures to the targeted compound get mixed in during the manufacturing process for intermediates. Just how can the targeted compound be extracted when this occurs? The patent this article reviews involves such an isomer mixture purification technology.
■ What you can learn from this article ✔ A technology for purifying high-purity 3-iodoaniline from an isomer mixture has been patented. ✔ By adding acid to turn liquid iodoaniline into a salt, recrystallization purification became possible. ✔ This purification technology was developed in response to an order from a foreign pharmaceutical manufacturer. ■ Recommended Articles ・ Hydrocarbon iodination: Aromatic compound iodination overview and reactions: Aromatic compound iodination reactions (1): Discussion series on bromination/iodination reactions 15 ・ Iodination of phenols, phenol ethers, anilines, and aniline-related compounds: Aromatic compound iodination reactions (2): Discussion series on bromination/iodination reactions 16 ・ Iodination of carboxylic acid, nitro compounds, and other inactive aromatic compounds: Aromatic compound iodination reactions (3): Discussion series on bromination/iodination reactions 17
contents
What are isomers?
For example, when using building blocks, you can put together many different structures depending on how you arrange them, even when using the same blocks. This is the same for chemical compounds that are made in the chemistry world. Compounds are formed by combining atoms, and different compounds can be made by changing the arrangement of the same exact atoms. Compounds made of the same atoms but arranged differently are called isomers (structural isomers).
Since isomers are structurally similar, isomers may simultaneously occur when producing a targeted compound if isomers for the targeted compound exist.
Challenges in making 3-iodoaniline
The patent being reviewed this time is for an invention of a purification method for obtaining 3-iodoaniline from a iodoaniline isomer mixture.
Aniline is a compound formed from a benzene ring and an amino group (-NH2) and is popular with such uses as dyes, rubber, and pharmaceuticals. Iodoaniline is formed by combining aniline and iodine, for which there exists three isomers that differ in the positional relationship between the NH2 group and iodine. When producing 3-iodoaniline, a small percentage of the solution will contain the 2-iodoaniline and 4-iodoaniline isomers, requiring the extraction of 3-iodoaniline from the isomer mix.
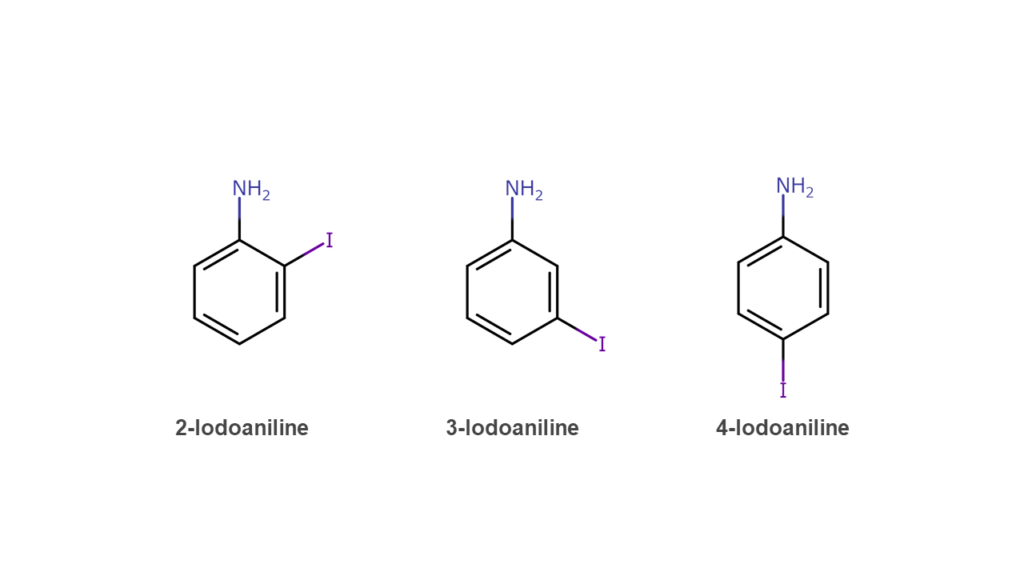
One might think, “Aren’t there any methods for producing higher percentage yields of 3-iodoaniline from the start?” Such methods exist, but are not suited for mass production due to cost and productivity issues. For that reason, the method in this patent approaches this problem by creating an iodoaniline isomer mix using a low-cost process and extracting 3-iodoaniline from the mix.
Generally, processes to purify a targeted compound from an isomer mix involves methods such as recrystallization (melting crude crystals, cooling the melted solution, and separating crystals of the targeted compound from the solution) and distillation (utilizing differing boiling points between the isomers to purify a liquid isomer mix). However, iodoaniline cannot be recrystallized as the compound is liquid at room temperature, and cannot easily be distilled as it is thermally sensitive. Research into just how purification could be made possible was conducted.

The certain chemical reaction that makes purification possible
The method developed this time involves adding an acid to an iodoaniline isomer mix to form a salt from the iodoaniline. This salt-containing solution is then recrystallized, and 3-iodoaniline salts are selectively separated.
The addition of hydrochloric acid to an iodoaniline isomer mix will yield the iodoaniline hydrochloride salts shown below. Salts are akin to common table salt (sodium chloride) and are solid crystals at room temperature. In short, this method takes iodoaniline, a liquid compound that cannot be recrystallized in its original state, and transforms it into a solid salt, enabling the use of recrystallization purification. This novel method can be thought of as the heart of this patent.
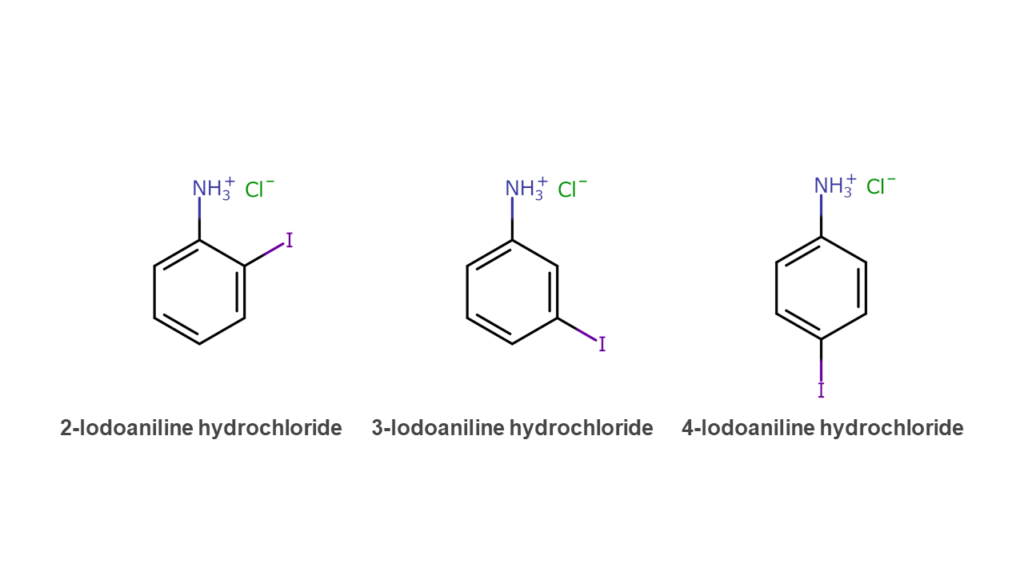
When hydrochloric acid is added to the isomer mix, each isomer salt is in a melted liquid state. Cooling this solution to approximately 0 to 5 degrees Celsius selectively recrystallizes 3-iodoaniline hydrochloride as it accounts for most of the solution content. In experiments, satisfactory results were achieved with 3-iodoaniline hydrochloride obtained at more than 99% purity. The resulting 3-iodoaniline hydrochloride obtained through recrystallization can be reverted to 3-iodoaniline with the use of a chemical base.
This method achieves low-cost production of high-purity 3-iodoaniline.
It began with an order from a non-Japanese pharmaceutical manufacturer
One of the core members behind the patent application for this technology is Hiroaki Miyahara, Pharmaceutical Team member of Research and Development. This purification method was developed after receiving an order from a non-Japanese pharmaceutical manufacturer to produce 3-iodoaniline.
Mr. Miyahara found that this purification method, created by Mr. Miyahara himself, may have patentability, and took charge of carrying out the entire patent process, from researching existing technologies to producing patent specification documents. The patent application was filed in 2012 and was subsequently accepted and registered without issue.
“Applying for and securing patent rights is extremely important for companies,” explains Mr. Miyahara, who we suspect will contribute to many more patent applications to come.